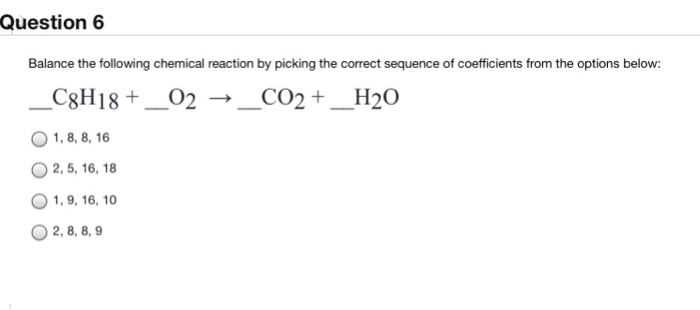
C8H18 +O2 =CO2 + H2O Balance ||Octane Complete Combustion Balanced Equation| C8H8 +O2 YieldsCO2,H2O - YouTube
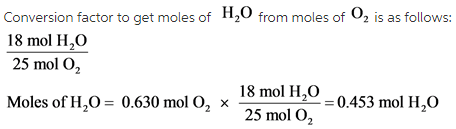
Determine the balanced chemical equation for this reaction. C8H18(g)+O2(g)→ CO2(g)+H2O(g) - Home Work Help - Learn CBSE Forum
When 10 g of CxHy is burned, 45.00 g of products are formed (CO2 and H2O combined), what is the empirical formula of the fuel (CxHy + O2→ CO2 + H2O)? - Quora

When octane (C8H18) is burned in air, it chemically reacts with oxygen gas ( O2) to produce carbon dioxide (CO2) and water (H2O). What mass of water is produced by the reaction of
C8H18 +O2 =CO2 + H2O Balance ||Octane Complete Combustion Balanced Equation| C8H8 +O2 YieldsCO2,H2O دیدئو dideo

SOLVED: SECTION 3: ANSWER ONE QUESTION FROM THIS SECTION. Balance the following chemical reactions. (2 Marks Each) C8H18 + O2 → CO2 + H2O C9H20 + O2 → CO2 + H2O +